- Overvew
- Table of Content
- Segmentation
- Request Sample
Market Definition
The gene editing technologies market is defined as the global economic sector encompassing the development, commercialization, and application of precision molecular tools used to deliberately alter the genomic sequences of living organisms. As of 2026, this market includes the revenue generated from the sale of specialized reagents, kits, and enzymes, as well as the software and contract services required to insert, delete, or replace DNA at specific locations within a genome. Unlike earlier forms of genetic engineering that relied on random insertion, the modern gene editing market is characterized by site-specific precision, enabling targeted modifications in human therapeutics, agricultural crop enhancement, and industrial biotechnology.
The technological scope of the market is primarily dominated by the CRISPR-Cas9 system, though it also includes established platforms such as Zinc Finger Nucleases (ZFNs) and Transcription Activator-Like Effector Nucleases (TALENs), as well as next-generation modalities like base editing and prime editing. These tools are utilized by a diverse range of end-users, including pharmaceutical and biotechnology companies, academic research institutes, and contract research organizations (CROs). The market’s reach extends across multiple functional areas, from cell line engineering and drug discovery to the development of curative therapies for rare genetic disorders and the creation of resilient, high-yield genetically modified crops.
In the current 2026 landscape, the market definition has expanded to include the infrastructure for non-viral delivery systems and AI-driven genomic design, reflecting a shift toward safer, more scalable clinical applications. It is increasingly defined by its transition from a research-heavy field to a commercially mature industry, driven by the rising prevalence of chronic genetic diseases and the global demand for precision medicine. Consequently, the market encompasses not just the biological scissors themselves, but the entire ecosystem of intellectual property, regulatory compliance services, and manufacturing technologies that enable the transformation of genetic code into viable commercial products.
Market Insights
The global gene editing technologies market is entering a phase of rapid commercial maturation in 2026, with a projected valuation exceeding USD12 billion. This growth is underpinned by a compound annual growth rate (CAGR) of approximately 16-19%, driven by a decisive shift from laboratory-based research to large-scale clinical and industrial applications. While North America continues to command the largest market share, exceeding 40% due to its robust biotech ecosystem and sustained capital investment, the Asia-Pacific region is emerging as the fastest-growing geographical segment. This regional surge is driven by expanding bio economies, localized manufacturing of reagents, and increasingly supportive regulatory frameworks for agricultural and therapeutic innovations.
A primary catalyst for this market expansion is the rising prevalence of chronic genetic disorders and the increasing clinical success of one-time curative therapies. Traditional symptomatic management is being steadily replaced by precision medicine, particularly for conditions like sickle cell disease, beta-thalassemia, and various oncology indications. CRISPR-Cas9 remains the dominant technology, capturing over 65% of the market share in 2026 due to its relative cost-effectiveness and ease of use. However, the market is diversifying with the rapid adoption of next-generation modalities such as base editing and prime editing, which offer higher precision with fewer off-target risks, thereby attracting significant venture capital and pharmaceutical partnerships.
The integration of Artificial Intelligence (AI) and Machine Learning (ML) has become a critical trend, revolutionizing how companies identify genomic targets and predict off-target effects. In 2026, AI-driven platforms are significantly reducing the R&D timeline for drug discovery and validating CRISPR-based diagnostics, which provide rapid, high-accuracy detection for infectious diseases. Beyond human health, the market is seeing a surge in agricultural biotechnology, where gene editing is utilized to develop climate-resilient, drought-tolerant, and nutritionally enhanced crops. This diversification into the non-therapeutic sector is providing a stable secondary revenue stream for technology providers and specialized contract research organizations (CROs).
Despite the bullish outlook, the market faces persistent structural challenges, most notably the high cost of clinical-grade manufacturing and the complexities of in vivo delivery systems. The technical hurdle of delivering molecular scissors safely into specific tissues remains a bottleneck for treating complex neurodegenerative and cardiovascular conditions. Furthermore, the industry is navigating a fragmented global regulatory landscape and ongoing ethical debates regarding germline editing. Consequently, the 2026 market is increasingly defined by strategic consolidation, where established players are acquiring specialized delivery technology firms to streamline the path from genomic design to commercial patient access.
Key Drivers
Clinical Success of One-Time Curative Therapies: The foremost driver of the market in 2026 is the decisive shift from chronic symptom management to one-time curative interventions for monogenic diseases. Following the landmark approvals of CRISPR-based treatments for sickle cell disease and beta-thalassemia, the clinical pipeline has expanded to include more complex indications such as hemophilia and hereditary blindness. These therapies offer a high-value proposition for both patients and healthcare systems by potentially eliminating the need for lifelong, multi-million-dollar supportive care. This curative model has attracted unprecedented levels of private equity and pharmaceutical investment, as stakeholders prioritize assets that can provide durable, long-term clinical benefits through a single administration.
Evolution of Precision Editing and Non-Viral Delivery: The rapid adoption of next-generation editing modalities, specifically base editing and prime editing, is significantly expanding the market’s addressable range. Unlike the first generation of CRISPR-Cas9, which creates double-stranded DNA breaks, these newer tools allow for the precise replacement of single nucleotides without cutting the DNA backbone, drastically reducing the risk of off-target mutations. Concurrently, the market is being driven by a transition toward non-viral delivery systems, such as lipid nanoparticles (LNPs) and advanced electroporation. These technologies address the historical bottlenecks of viral vectors, such as immunogenicity and high manufacturing costs, enabling safer, more scalable, and cost-effective production of gene-edited cell therapies.
Key Challenges
Prohibitive Commercialization and Access Costs: Despite the scientific breakthroughs, the staggering cost of gene-edited therapies remains a primary market challenge in 2026. The price tags for these treatments often exceed USD2 million to USD3 million per patient, creating a reimbursement gap that strains even the most robust national health insurance systems. This financial barrier is compounded by the high cost of clinical-grade manufacturing and the specialized infrastructure required for ex vivo cell handling. Consequently, there is a significant disparity in market access, where life-saving genomic medicines are primarily restricted to high-income regions, limiting the global volume of patients and hindering the market’s expansion into emerging bioeconomies.
Ethical Ambiguity and Regulatory Fragmentation: The market faces a complex and often contradictory global regulatory landscape, particularly regarding the ethical boundaries of genomic modification. While many nations have established clear pathways for somatic cell editing (non-heritable), there remains a profound lack of international consensus on germline editing (heritable) and enhancement technologies. This fragmentation creates uncertainty for multinational biotech firms, who must navigate varying safety standards, patent disputes, and bioethical moratoria that differ by jurisdiction. This regulatory patchwork not only slows down cross-border clinical trials but also invites public skepticism, as the industry struggles to establish a unified ethical framework for designing life.
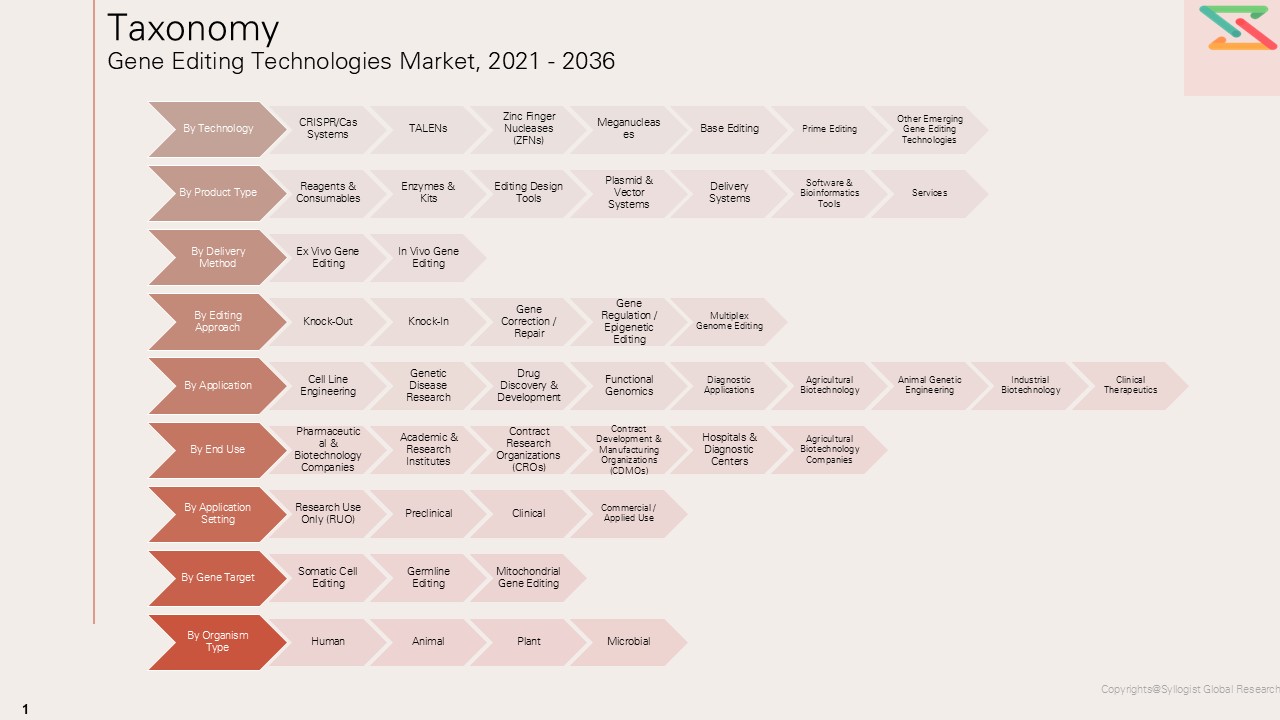
Market Segmentation
- Segmentation By Technology
- CRISPR/Cas Systems
- TALENs
- Zinc Finger Nucleases (ZFNs)
- Meganucleases
- Base Editing
- Prime Editing
- Other Emerging Gene Editing Technologies
- Segmentation By Product Type
- Reagents & Consumables
- Enzymes & Kits
- Editing Design Tools
- Plasmid & Vector Systems
- Delivery Systems
- Software & Bioinformatics Tools
- Services
- Market Size & By Delivery Method
- Ex Vivo Gene Editing
- In Vivo Gene Editing
- Segmentation By Editing Approach
- Knock-Out
- Knock-In
- Gene Correction / Repair
- Gene Regulation / Epigenetic Editing
- Multiplex Genome Editing
- Segmentation By Application
- Cell Line Engineering
- Genetic Disease Research
- Drug Discovery & Development
- Functional Genomics
- Clinical Therapeutics
- Agricultural Biotechnology
- Animal Genetic Engineering
- Industrial Biotechnology
- Diagnostic Applications
- Segmentation By End Use
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Contract Research Organizations (CROs)
- Contract Development & Manufacturing Organizations (CDMOs)
- Hospitals & Diagnostic Centers
- Agricultural Biotechnology Companies
- Segmentation By Application Setting
- Research Use Only (RUO)
- Preclinical
- Clinical
- Commercial / Applied Use
- Segmentation By Gene Target
- Somatic Cell Editing
- Germline Editing
- Mitochondrial Gene Editing
- Segmentation By Organism Type
- Human
- Animal
- Plant
- Microbial
All market revenues are presented in USD
Historical Year: 2021–2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027–2036
Key Questions this Study Will Answer
- What are the critical market metrics and forward-looking projections for the Global Gene Editing Technologies Market, including revenue size, therapeutic/diagnostic manufacturing capacity, unit volume (kits/assays/systems), and average pricing by technology type (CRISPR-Cas, Base Editing, Prime Editing, TALENs, ZFNs), scale of operation (Research-grade, Clinical-grade/GMP, Industrial-scale), delivery modality (Viral Vectors, Lipid Nanoparticles, Electroporation, Biolistics), configuration (Ex vivo, In vivo, In vitro/Cell-free), application (Gene Knock-out, Gene Knock-in, Epigenetic Editing, Functional Genomics, Site-Directed Mutagenesis), end-use industry (Human Therapeutics, Agriculture, Animal Health, Synthetic Biology, Diagnostics, Forensics), and commercial structure (Product-based, CRO/Service-based, IP Licensing, Royalty/Milestone-based)?
- How do supply–demand fundamentals vary across key regions and major consuming industries, and what role do genomic research funding, disease prevalence (monogenic/oncology), healthcare expenditure, bio-security policies, regulatory fast-track designations, and laboratory infrastructure gaps play in shaping regional market competitiveness, alongside local capability in GMP facility construction, enzyme/reagent synthesis, high-fidelity gRNA fabrication, bioinformatics/AI-driven design, clinical trial management, and downstream specialty pharmacy connectivity, as well as procurement structures across direct asset ownership, long-term master service agreements (MSAs), EPC (Engineering, Procurement, and Commissioning) for labs, and collaborative R&D arrangements?
- In what ways are raw material, equipment, and biological component price volatility, along with engineering and cleanroom construction lead-time constraints, covering high-fidelity enzymes (Cas9/Cas12), synthetic guide RNA, viral vector plasmids, LNP components (ionizable lipids), automated liquid handlers, NGS sequencers, specialized incubators, and high-purity chemicals, influencing project CAPEX, per-treatment costs, developer margins, FDA/EMA submission timelines, and lifecycle economics, especially for projects requiring complex multiplex editing, non-viral delivery vehicles, off-target monitoring systems, and harsh-environment (ultra-low temperature) cold chain logistics?
- Who are the leading global and regional Gene Editing developers, CDMOs, equipment providers, and technology licensors, and how do they benchmark across key performance dimensions including editing efficiency (percentage), off-target toxicity profiles, throughput/scale, uptime and batch consistency, delivery versatility, turnaround time for customized cell lines, IP portfolio strength, relocation/tech-transfer capability, safety performance, regulatory track record, digital/AI monitoring capability, and value-added offerings such as integrated bench-to-bedside solutions, modular GMP deployment, and flexible licensing/royalty structures?
- What strategic insights emerge from primary discussions with biotech developers, pharmaceutical executives, clinical researchers, agricultural scientists, genomic investors, and regulatory consultants regarding demand shifts toward high-precision tools (Base/Prime editing), specification changes driven by smaller-scale niche orphan disease opportunities, gas-to-power equivalents in genomics (e.g., cell-based manufacturing), commercialization of stranded genomic data, decarbonization/sustainability expectations in bio-industrial applications, and regional sourcing strategies for critical biological components, preferences between in-house development versus outsourced CDMO models, and key investment criteria such as clinical durability, speed-to-market, regulatory de-risking, editing precision, reliability, manufacturing intensity, and counterparty intellectual property strength?
- Product Definition
- Research Methodology
- Research Design & Framework
- Overall Research Approach: Descriptive, Exploratory & Quantitative Mixed-Method Design
- Market Definition & Scope Boundaries: What is Included and Excluded
- Segmentation Framework
- Key Research Assumptions & Limitations
- Secondary Research
- Primary Research Design & Execution
- Data Triangulation & Validation
- Market Sizing & Forecasting Methodology
- Competitive Intelligence Methodology
- Quality Assurance & Peer Review
- Definitions, Abbreviations & Data Notes
- Research Design & Framework
- Executive Summary
- Market Snapshot & Headline Numbers
- Key Findings & Research Highlights
- Market Dynamics
- Regional Market Summary
- Competitive Landscape Snapshot
- Technology & Innovation Highlights
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Challenges
- Porter’s Five Forces Analysis
- PESTLE Analysis
- Market Trends & Developments
- Emerging Trends
- Technological Developments
- Regulatory & Policy Changes
- Supply Chain & Sourcing Trends
- Consumer & Demand Trends
- Investment & Funding Activity
- Sustainability & ESG Trends
- Geographic & Regional Trends
- Risk Assessment Framework
- Political / Geopolitical Risk
- Fuel & Component Supply Risk
- Environmental and Regulatory Risk
- Financial / Market Risk
- Regulatory Framework & Standards
- Global Gene Editing Technologies Market Outlook
- Market Size & Forecast by Value
- Market Size & Forecast by Technology
- CRISPR/Cas Systems
- TALENs
- Zinc Finger Nucleases (ZFNs)
- Meganucleases
- Base Editing
- Prime Editing
- Other Emerging Gene Editing Technologies
- Market Size & Forecast by Product Type
- Reagents & Consumables
- Enzymes & Kits
- Editing Design Tools
- Plasmid & Vector Systems
- Delivery Systems
- Software & Bioinformatics Tools
- Services
- Market Size & By Delivery Method
- Ex Vivo Gene Editing
- In Vivo Gene Editing
- Market Size & Forecast by Editing Approach
- Knock-Out
- Knock-In
- Gene Correction / Repair
- Gene Regulation / Epigenetic Editing
- Multiplex Genome Editing
- Market Size & Forecast by Application
- Cell Line Engineering
- Genetic Disease Research
- Drug Discovery & Development
- Functional Genomics
- Clinical Therapeutics
- Agricultural Biotechnology
- Animal Genetic Engineering
- Industrial Biotechnology
- Diagnostic Applications
- Market Size & Forecast by End Use
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Contract Research Organizations (CROs)
- Contract Development & Manufacturing Organizations (CDMOs)
- Hospitals & Diagnostic Centers
- Agricultural Biotechnology Companies
- Market Size & Forecast by Application Setting
- Research Use Only (RUO)
- Preclinical
- Clinical
- Commercial / Applied Use
- Market Size & Forecast by Gene Target
- Somatic Cell Editing
- Germline Editing
- Mitochondrial Gene Editing
- Market Size & Forecast by Organism Type
- Human
- Animal
- Plant
- Microbial
- Asia-Pacific Genset Market Outlook
- Market Size & Forecast
- By Value
- By Technology
- By Product Type
- By Delivery Method
- By Editing Approach
- By Application
- By End Use
- By Application Setting
- By Gene Target
- By Organism Type
- Market Size & Forecast
- Europe Genset Market Outlook
- Market Size & Forecast
- By Value
- By Technology
- By Product Type
- By Delivery Method
- By Editing Approach
- By Application
- By End Use
- By Application Setting
- By Gene Target
- By Organism Type
- Market Size & Forecast
- North America Genset Market Outlook
- Market Size & Forecast
- By Value
- By Technology
- By Product Type
- By Delivery Method
- By Editing Approach
- By Application
- By End Use
- By Application Setting
- By Gene Target
- By Organism Type
- Market Size & Forecast
- Latin America Genset Market Outlook
- Market Size & Forecast
- By Value
- By Technology
- By Product Type
- By Delivery Method
- By Editing Approach
- By Application
- By End Use
- By Application Setting
- By Gene Target
- By Organism Type
- Market Size & Forecast
- Middle East & Africa Genset Market Outlook
- Market Size & Forecast
- By Value
- By Technology
- By Product Type
- By Delivery Method
- By Editing Approach
- By Application
- By End Use
- By Application Setting
- By Gene Target
- By Organism Type
- Market Size & Forecast
- Country Wise* Genset Market Outlook
- Market Size & Forecast
- By Value
- By Technology
- By Product Type
- By Delivery Method
- By Editing Approach
- By Application
- By End Use
- By Application Setting
- By Gene Target
- By Organism Type
- Market Size & Forecast
*Countries Analyzed in the Syllogist Global Research Portfolio: United States, Canada, Germany, United Kingdom, France, Italy, Spain, Netherlands, Switzerland, China, Japan, India, South Korea, Australia, Brazil, Mexico, Saudi Arabia, South Africa, UAE
- Technology Landscape & Innovation Analysis
- Evolution of Programmable Nucleases
- Precision Editing Breakthroughs
- Delivery System Innovations
- AI & Digital Transformation in Gene Design
- Value Chain & Supply Chain Analysis
- Pricing Analysis
- Sustainability & Energy Efficiency
- Green Laboratory Operations
- Sustainable Bioprocessing Technologies
- Gene Editing as a Sustainability Tool
- Competitive Landscape
- Market Structure & Concentration
- Market Consolidation Level (Fragmented Vs Consolidated)
- Top 5 Players Market Share
- HHI (Herfindahl–Hirschman Index) Concentration Analysis
- Competitive Intensity Map
- Player Classification
- Market Leaders
- Strong Challengers
- Specialist / Niche Players
- Emerging Players
- Regional Players
- Competitive Analysis Frameworks
- Market Share Analysis
- Company Profile
- Company Overview & HQ
- Products & Solutions Portfolio
- Overall Revenue & Segmental Revenue
- Geographic Presence
- Recent Developments (M&A, Launches, Partnerships)
- SWOT Analysis
- Strategic Focus Areas
- Competitive Positioning Map
- Market Structure & Concentration
- Strategic Output
- Market Opportunity Matrix
- White Space Opportunity Analysis
- Strategic Recommendations
- For Tool Providers
- For Therapeutic Developers
- For Investors
- Strategic Priority Matrix & Roadmap
- Near-term (2025–2028)
- Mid-term (2029–2032)
- Long-term (2033–2036)